Preface
Novapura had an independent company conduct a survey on the topic of “disinfection in hospitals” in May 2022. Institutions in Switzerland were surveyed. (Compare also the blog from 15.7.22)
Questions and answers
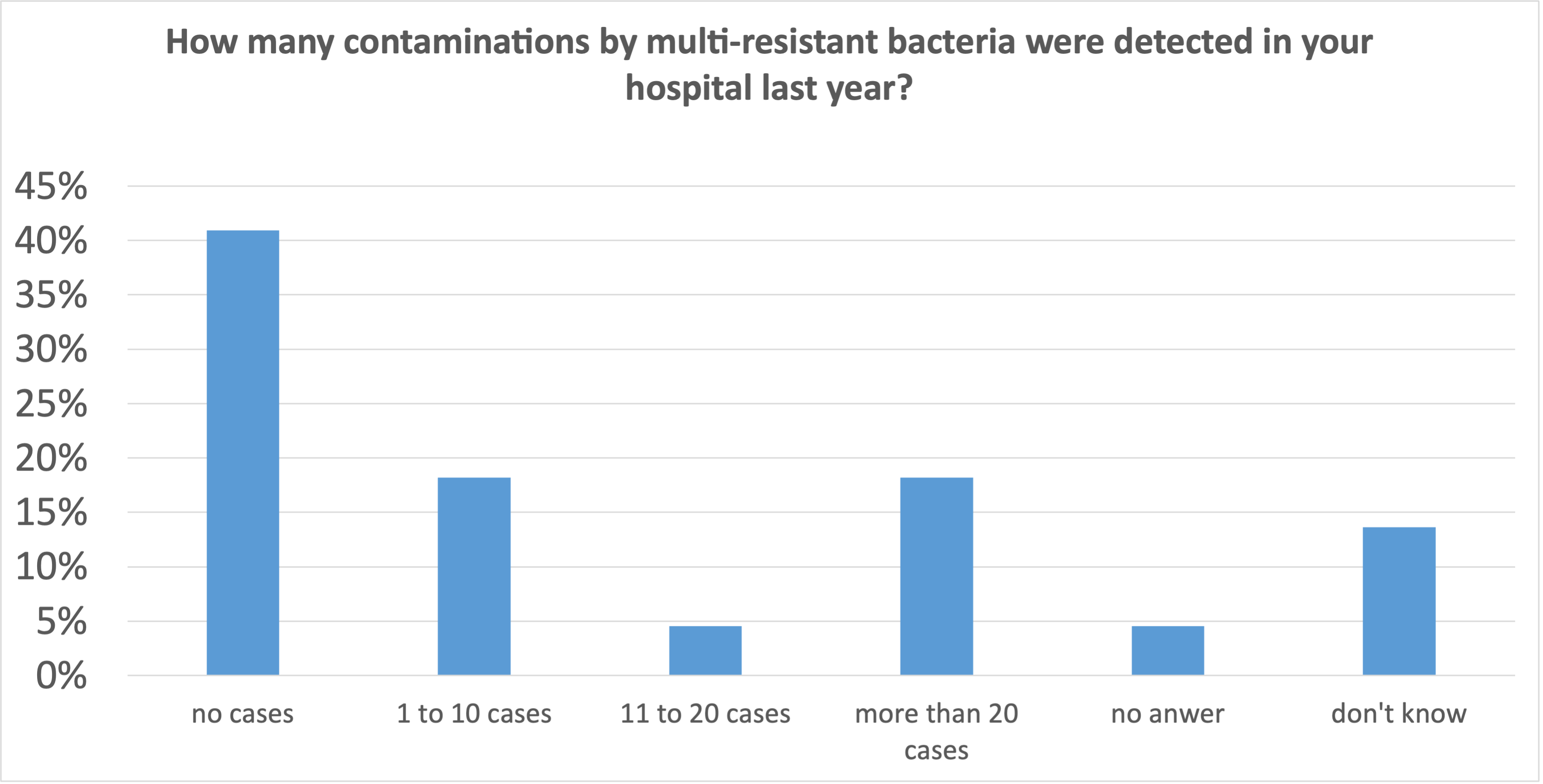
- 41% of the institutions had no cases to fight.
- 41% of the institutions had more than one case to fight, 18% of the institutions had to deal with more than 20 cases per year.
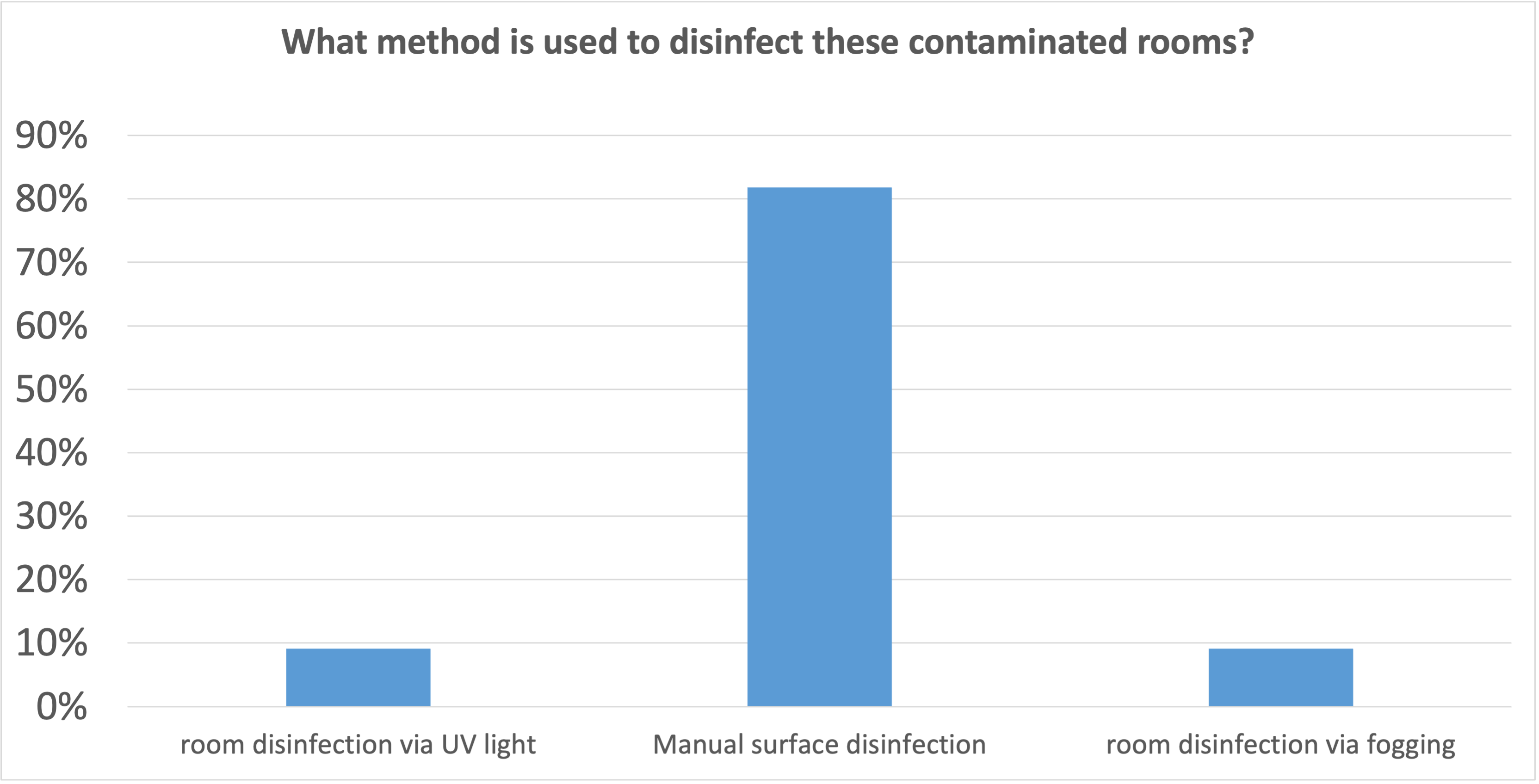
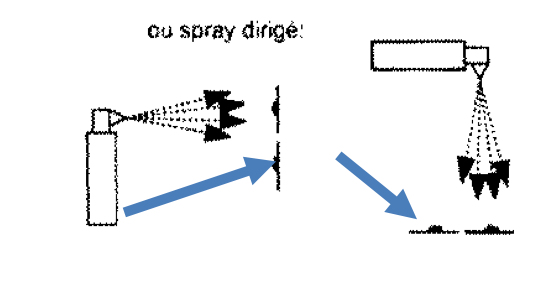
In more than 80% of the institutions, manual surface disinfection is also used in these cases. In other words, disinfection is done by hand. Despite obvious deficiencies, such as
- lack of process safety, i.e. the quality of the disinfection is left to the good will of the person carrying it out
- the process is labour-intensive and takes a long time
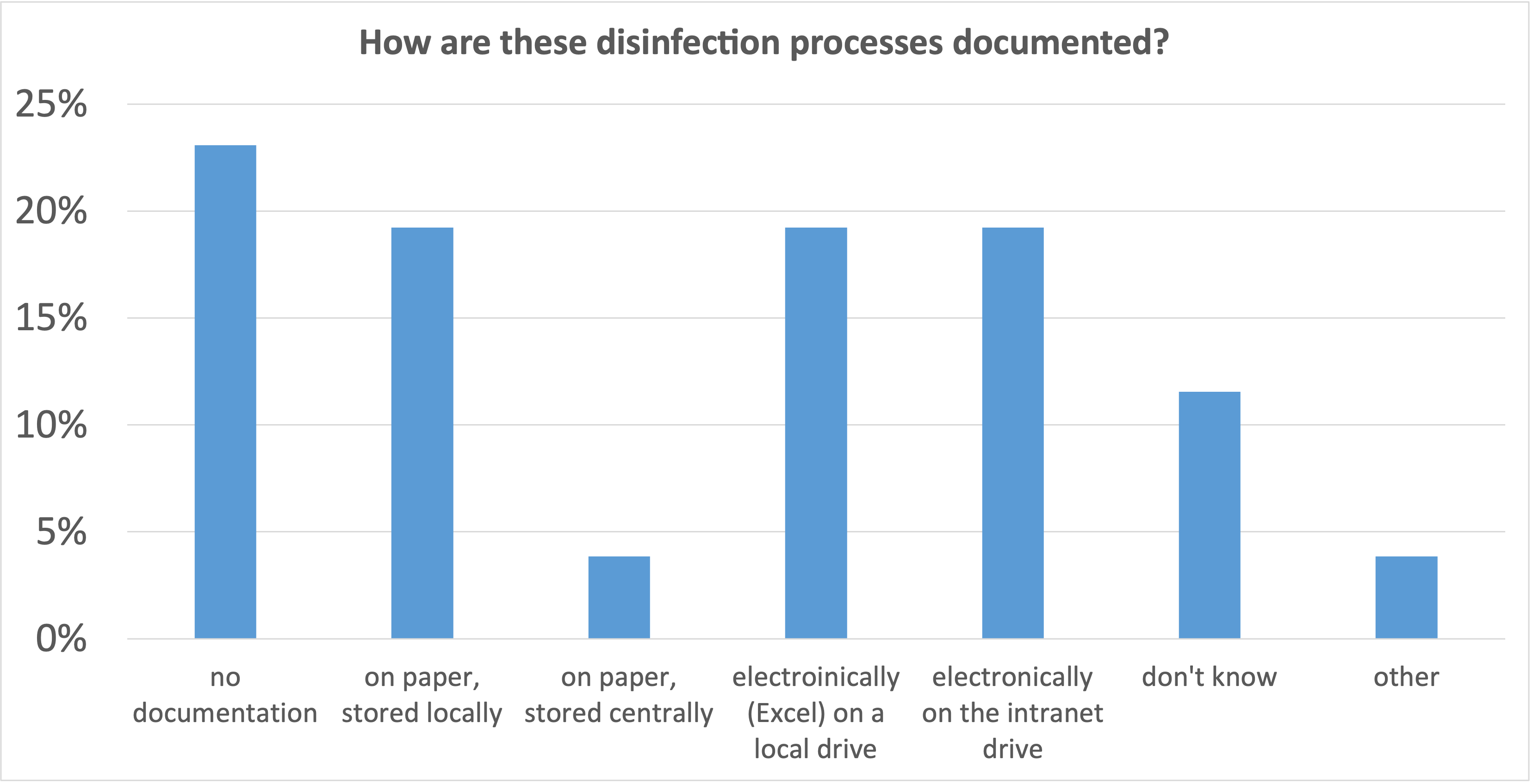
23% of the institutions report that the disinfection process is not documented at all, in 62% of the institutions it is documented, but only in just under 20% of the cases there is a central filing system.
In order to make a process safe and reliable, a clear documentation is indispensable. It is only through the evaluation of this documentation that errors can be detected and corrected. Over time, the process becomes more stable and the quality improves sustainably.
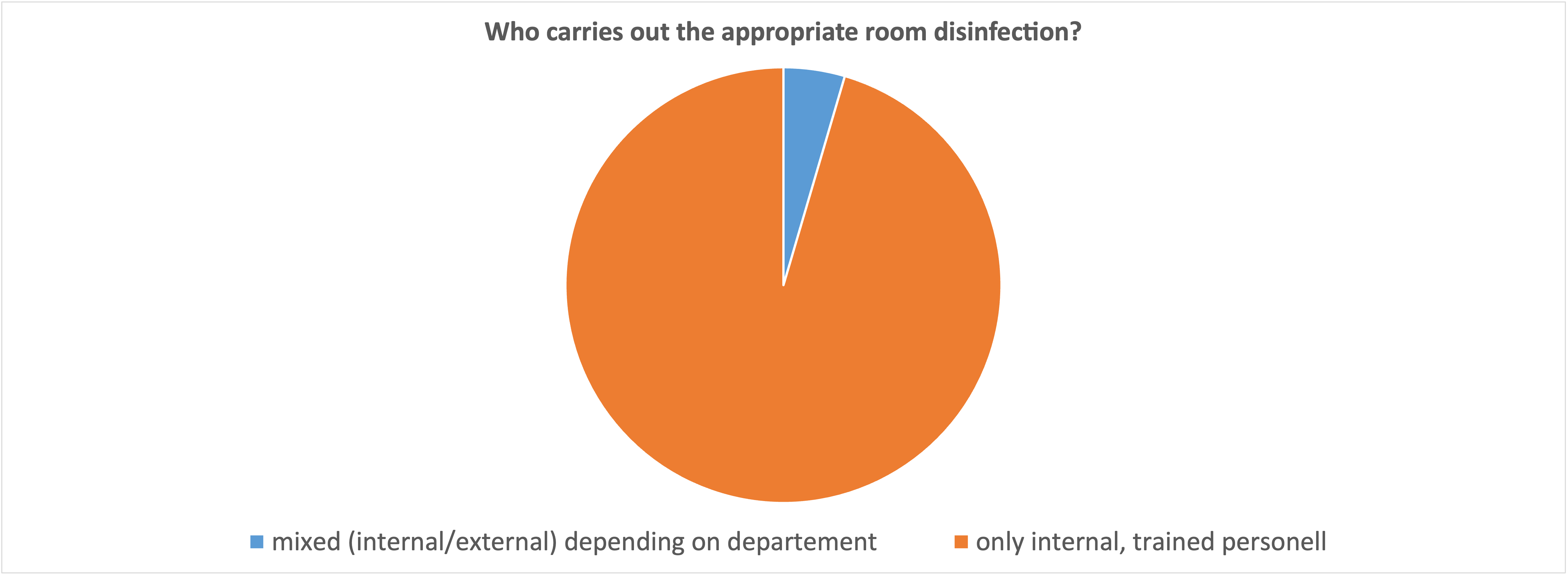
What is astonishing from our point of view is the answer to the question of who carries out the decontamination after an attack of multi-resistant bacteria: It is essentially internally trained staff. In times of scarce human resources, it is worth considering whether it is not better to outsource this process completely. A professional, external service provider is faster and more experienced than internal staff, who (according to this survey) only carry out decontamination every second week at most. Added to this is the experience with personal protective equipment, which requires special attention when carrying out such complex and potentially dangerous work.
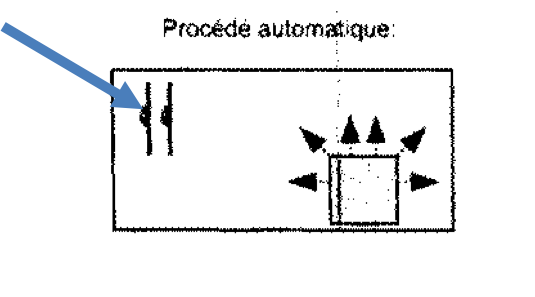
An alternative is the use of automatic systems, which are very easy to operate and require a minimum of personal protective equipment. This can massively increase the efficiency of the internal staff, because the systems work autonomously; during this time the operator can pursue another task.
Biggest challenge in today’s disinfection practice?
The following three key issues have been identified as the biggest challenge:
- The process is personnel and cost intensive
- Lack of records of disinfections performed
- Risk of infection and spread in the building
Findings
We see a lot of optimisation potential in the disinfection of rooms when they need to be decontaminated.
Automatic disinfection versus scrubbing and wiping disinfection by hand.
careful use of human resources through automation or consistent outsourcing of the process
stringent and consistent documentation versus decentralised, sometimes paper-based documentation. Learning cycles can be shortened, which benefits the staff and the patients.
Digitalisation must also find its way into this process.